

Women in Science: PCR in Galapagos Giant Tortoises
Gislayne, a biotechnologist from Galapagos explores the use of PCR in work to assess the health of Endangered giant tortoises.
Gislayne Mendoza is a biotechnologist from Galapagos working with the Charles Darwin Foundation (CDF) and funded by Galapagos Conservation Trust (GCT). In this blog, she explores the use of a specific scientific technique called Polymerase Chain Reaction technique (PCR) in fascinating work to assess the health of Endangered giant tortoises as part of our Giant Tortoise Movement Ecology Programme (GTMEP).

Ainoa Nieto prepares materials to collect biological samples from Alcedo Volcano giant tortoises while Freddy Cabrera takes morphometrics. Photo © Juan Manuel García, CDF
“In the warm month of May 2021, I started working at the Charles Darwin Research Station with GTMEP. As a Galapagueña, this has been a great opportunity to return home after finishing my undergraduate degree in Biotechnology Engineering at the Universidad San Francisco de Quito and to contribute to the study and conservation of our iconic giant tortoises.
I started working with Dr. Ainoa Nieto, Principal Investigator of GTMEP. Under her mentorship, we continued working in the molecular biology area, which had been stopped during the COVID-19 pandemic. My job was to detect and analyze specific viruses in Galapagos tortoises using the Polymerase Chain Reaction technique (PCR). But … what actually is a PCR?
PCR is a technique that has been used since Dr. Kary Mullis discovered it in 1990, but it has become world-renowned in the wake of the recent pandemic. Despite the fact that almost everyone on the planet has heard this word in the last two years, or even recently undergone a PCR, it is very likely that many do not know exactly what it is or why it is so important for disease diagnosis. This technique allows millions of copies of a specific DNA (deoxyribonucleic acid) fragment of a living organism to be obtained from a small biological sample. DNA, as you know, is where the genetic material of any species is found. These exact copies allow us to identify the DNA of that disease or pathogen that we are studying. For this reason, by analyzing the results, we can obtain important information such as, in our case, whether or not there are certain viruses in the Galapagos giant tortoises. This technique is very useful because it can be utilized to perform other analyses, such as paternity testing, forensic analysis, and detection of illegal wildlife trafficking, etc.
Before the PCR was invented studying DNA was very complicated because this structure contains a large amount of genetic information and detecting or isolating specific genes was a challenge. In fact, a decade before its discovery, gene cloning was the latest technology used by scientists to duplicate genetic material, but the work was laborious and very expensive. Since its creation just 32 years ago, the PCR is here to stay as a very useful technological tool, since it has managed to position itself in the field of science to this day and we have this technique to thank for the ability to detect SARS-CoV-2 in humans.
What do we need to carry out a PCR?
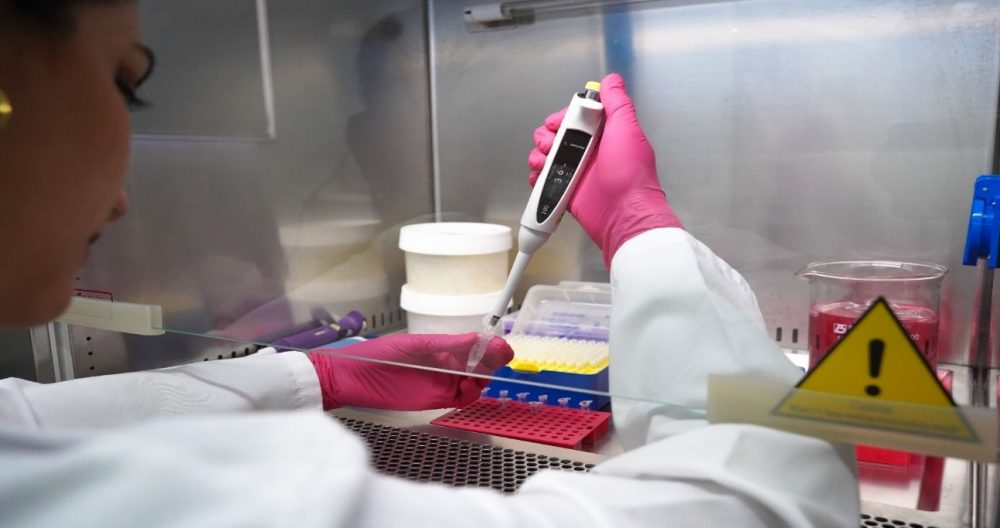
Gislayne Mendoza performing a PCR. In this step, she adds all “ingredients” into an Eppendorf tube with exact measurements previously calculated (this mixture is known as Master Mix). Photo © Rashid Cruz, CDF.
Carrying out a PCR is very similar to following a kitchen recipe, we need ‘ingredients’, measurements, exact times and ideal temperatures to obtain our result. Among the ingredients is, the “template” DNA fragment that will give us the genetic information that we need to “copy”. Here we can remember what we were taught in biology classes at school about this ladder-shaped molecule turned in helix format, which is part of all living beings on Planet Earth. In addition, we need primers or oligonucleotides, which are short DNA sequences specifically designed to help us find the region we want to study, and starting point to generate new DNA copies. To complete our list, we need a few important ingredients to extract and copy our DNA fragment, such as the buffer solution, dNTPs, Taq Polymerase enzyme and Magnesium ions (Wages, 2005).
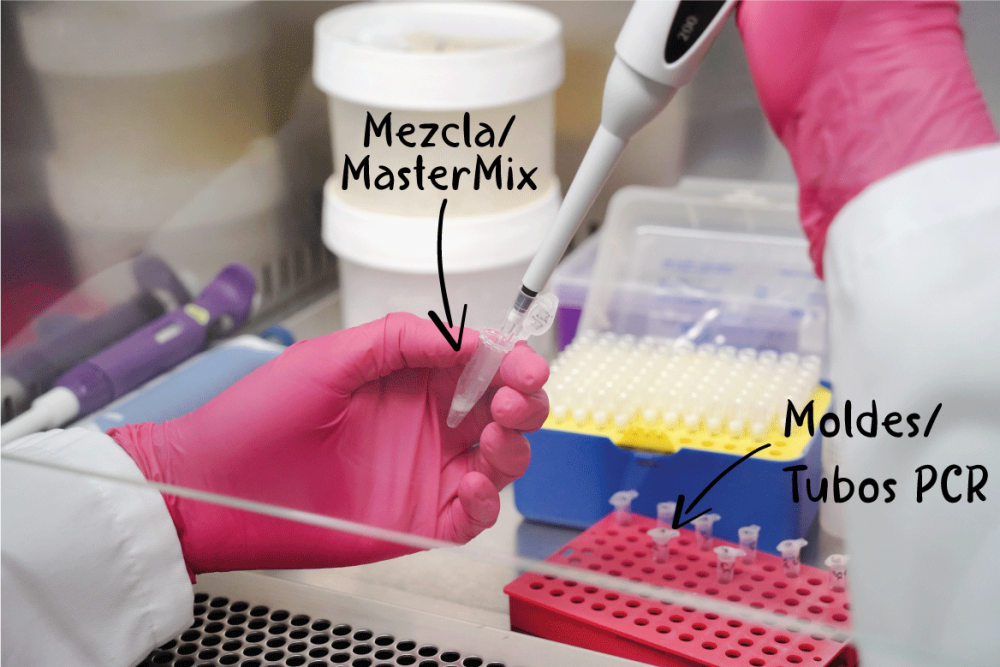
Once we have all our ingredients, we calculate the appropriate proportions, depending on the number of samples we want to analyze. Something similar to the quantities that we put when we prepare a cake and measure the ingredients according to the number of people. When we get our exact mix of ingredients, we dispense it into small tubes and take them to our ‘oven’, known as a thermocycler. This is a device that raises and lowers the temperature according to the programming indicated. This last step is essential to obtain the result of our long-awaited ‘cake’, since we need several cycles with ideal temperatures for the DNA chain to open, new fragments to be synthesized in each cycle and finally, those chains to close.

Gislayne Mendoza placing the sample in the thermocycler (our ‘oven’). Photo © Rashid Cruz, CDF.
To visualize the DNA fragments that interest us, another technique with a galactic name, called Electrophoresis is used. This is a laboratory technique that is responsible for separating DNA molecules with the help of an electric current. For this procedure, we need a device that contains a negative charge at one end and a positive charge at the other. Therefore, when inserting DNA molecules (which have a negative charge), they will migrate to the positive side, allowing DNA fragments to be detected in the form of bands, using a contrast with ultraviolet light (Michov, 2020). In this way, we can determine if the samples we are studying are positive for a certain virus, thanks to the presence of these bands, as shown in the following image:
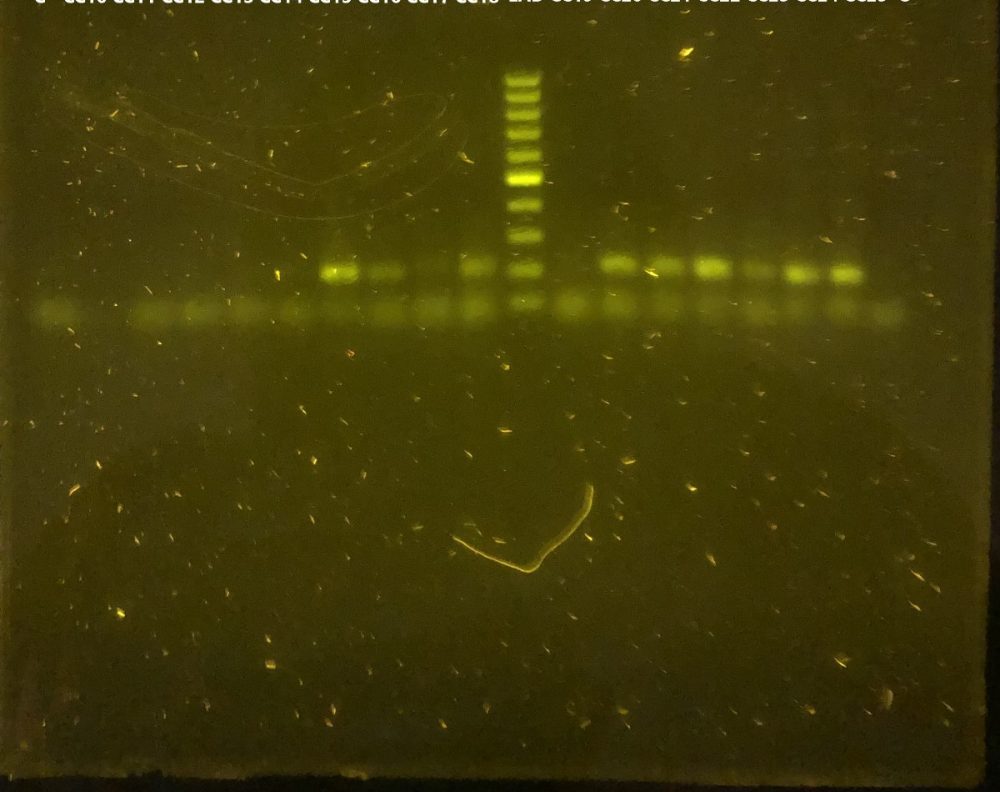
Agarose gel showing bands compatible with positive results of adenovirus from Galapagos tortoises at the Charles Darwin Research Station Fisher Laboratory. Photo © Gislayne Mendoza Alcivar, CDF
Within the GTMEP, the use of PCR has allowed us to describe four new viruses in giant tortoises: two adenoviruses and two herpesviruses. Although everything seems to indicate that these viruses are endemic to the islands, that is, they have evolved with their host (giant tortoises) over time, it is also suggested that these viruses could cause disease and leave long-term illness, especially in animals suffering from immunosuppression or stress from captivity or transport.

Ainoa Nieto providing instructions to Anne Guezou and Freddy Cabrera. An adult tortoise can be seen next to Ainoa, apparently, also paying attention to the instructions. Española Island. Photo: Juan Manual García, CDF.
The use of techniques such as PCR can help us better understand these risks. We can only conserve what we know, so it is important to continue this study in all species of Galapagos tortoise to catalogue the infectious agents that are present, and support the creation of prevention, control and surveillance plans for these diseases, so we can preserve these centuries-old species that are at risk. Thanks to my participation in this project, I have had the opportunity to gain theoretical and practical experience, both with new laboratory techniques to analyze the health of tortoises, working in the field and supporting different investigations within the program. In the laboratory, just like in the kitchen, we do not always get perfect results. With a lot of patience, I have been able to experiment and try different ‘ingredients’ to perfect and solve every detail, until I get ‘the perfect cake'”.

Gislayne Mendoza taking morphometric measurements of a giant tortoise. Photo © Karina Ramón, CDF.
This programme is a multi-institutional collaboration among the Charles Darwin Foundation, the Max Planck Institute of Ornithology, the Galapagos National Park, Saint Louis Zoo Institute for Conservation Medicine, the Houston Zoo and Galapagos Conservation Trust.
How to help Galapagos giant tortoises?
Donate towards the GTMEP programme or adopt a giant tortoise today! Alternatively, become a GCT member to commit long-term support and in return, receive our biannual magazine for updates from this project and others.
This blog was originally posted on the Charles Darwin Foundation website here and has been edited slightly for our website.
Related articles


Gills Club: Empowering young women in Galapagos

Isabel Cooper: A pioneering woman in Galapagos


